Glycol
Glycol is any of a class of organic compounds belonging to the alcohol family; in the molecule of a glycol, two hydroxyls (―OH) groups are attached to different carbon atoms. The term is often applied to the simplest member of the class, ethylene glycol.
Ethylene glycol is a colorless, oily liquid possessing a sweet taste and mild odor. It is produced commercially from ethylene oxide, which is obtained from ethylene. Ethylene glycol is widely used as antifreeze in automobile cooling systems and manufactures human-made fibers, low-freezing explosives, and brake fluid. Ethylene glycol and some of its derivatives are mildly toxic.
Propylene glycol, also called 1,2-propanediol, resembles ethylene glycol in its physical properties. Unlike ethylene glycol, however, propylene glycol is not toxic and is used extensively in foods, cosmetics, and oral hygiene products as a solvent, preservative, and moisture-retaining agent. Propylene glycol is manufactured in large amounts from propylene oxide, which is obtained from propylene.
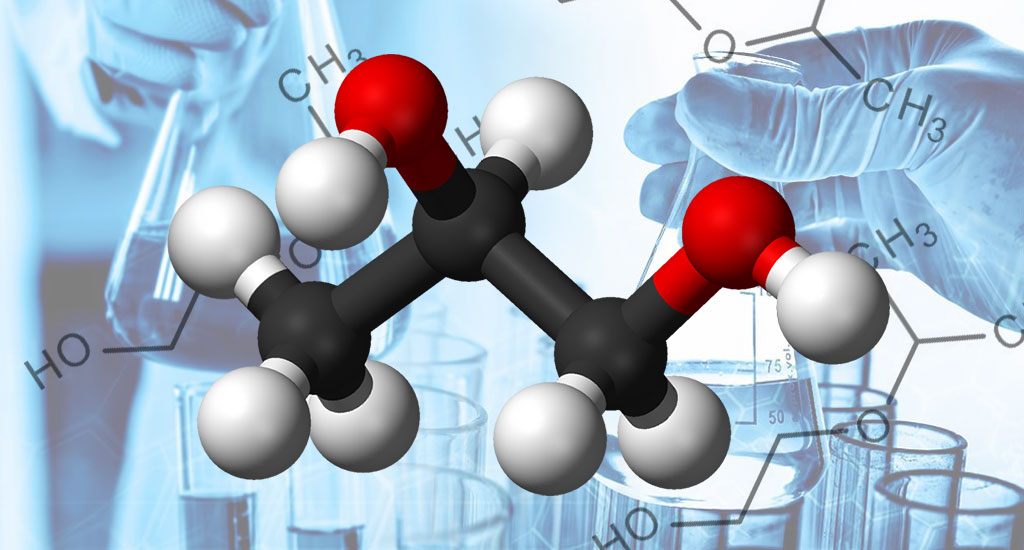
MEG - Monoethylene Glycol
Monoethylene glycol is obtained from the reaction of ethylene oxide and water. It is a clear, transparent, odorless liquid that can be mixed with water. Monoethylene glycol (MEG) is the simplest combination of glycols. And is one of the most common organic compounds with antifreeze and antifreeze properties.
Benefits
Moisture absorber
prevents freezing and freezing of water.
Applications
Used as a raw material for the preparation of polymers such as polyesters and polyethylene terephthalate
Application in antifreeze formulations in cars
Applied as a cooling and heat transfer facilitator in air conditioning systems
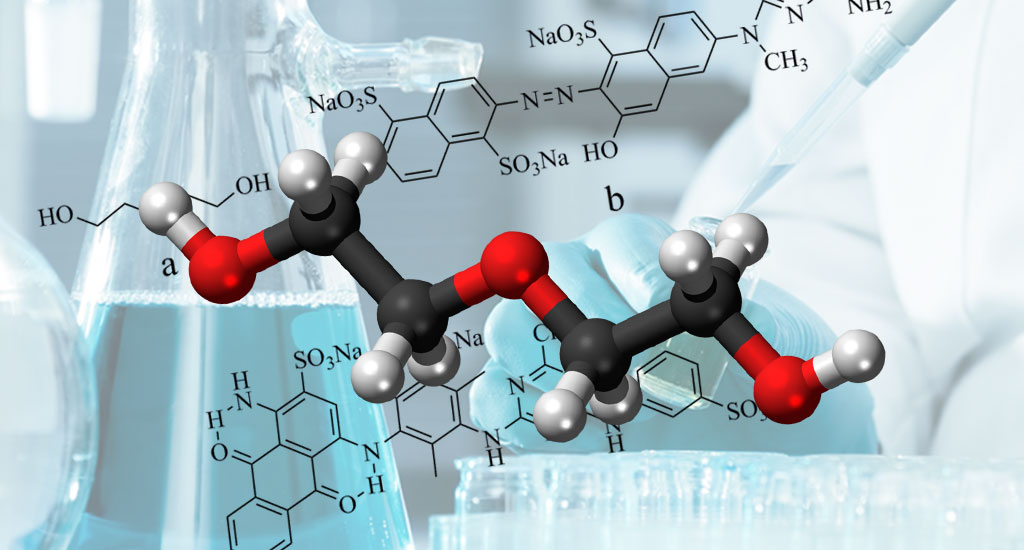
DEG - Diethylene Glycol
Diethylene glycol (DEG) is one of the most common organic solvents, covering a wide range of industrial applications. This compound can dissolve in other organic compounds. It is completely soluble in water. It is miscible with many organic solvents. It is miscible with water, ethanol, acetone, ether, and ethylene glycol.
Benefits
Organic solvent
Excellent moisturizing property
Applications
- the raw material for saturated polyester resins, unsaturated polyurethane resins, and plastics.
- the raw material for polyurethane and polyester
- thinner in the printing ink and textile industry
- thinner in the adhesive
- cosmetic industry
- metalworking fluid
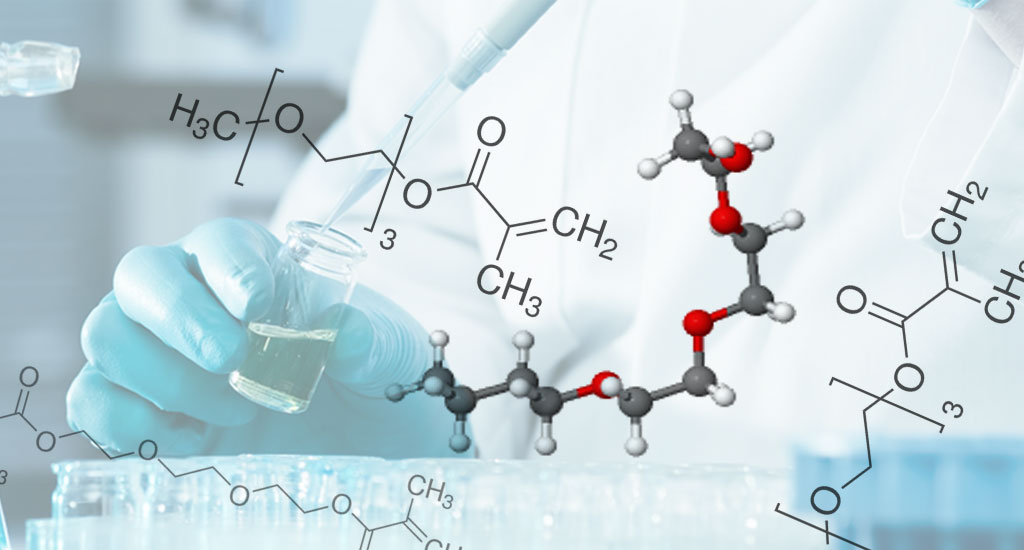
TEG - Triethylene Glycols
It is a colorless, viscous liquid with a low odor. Non-flammable, almost toxic, and harmless. The main application of this material is in the preparation of vinyl polymer as a plasticizer.
Benefits
Moisture absorber
air conditioner and lubricant for the production of many synthetic and natural fibers.
Applications
cosmetics
conditioners
shampoos
dyes in the textile industry
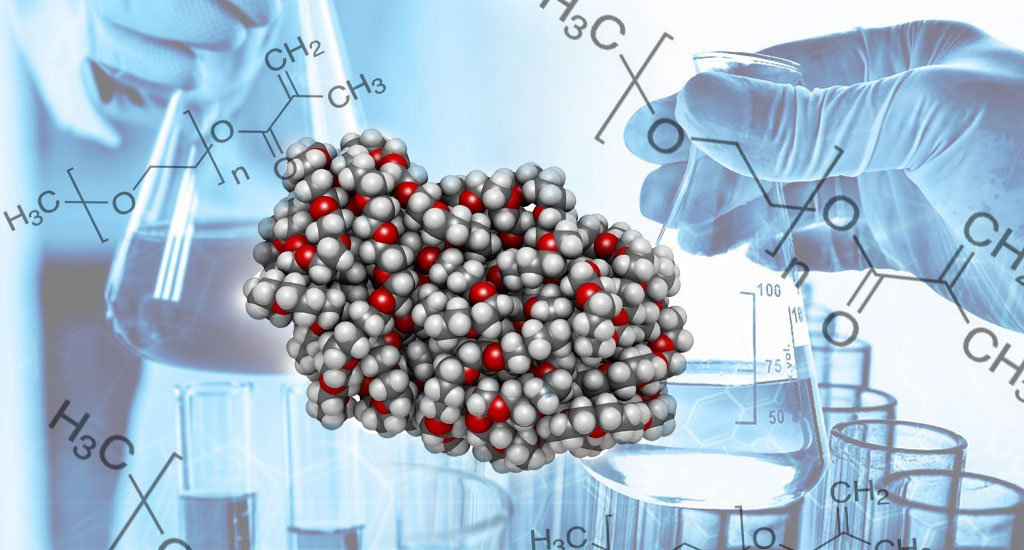
PEG - Polyethylene Glycols
Polyethylene glycols (PEGs) are a family of water-soluble linear polymers formed by the addition reaction of ethylene oxide (EO) with mono ethylene glycols (MEG) or diethylene glycol. A generalized formula for polyethylene glycol: H(OCH2CH2) n OH N: Average number of repeating ethylene oxide groups. Many grades of PEG represent them according to their average molecular weight. For example, PEG 400 consists of a dispersion of polymers of varying molecular weights, corresponding to the approximate average number of EO groups (n) of 400 repeats. Polyethylene glycols are available in average molecular weights ranging from 200 to 8000; this wide range provides flexibility in choosing features to meet the needs of many different applications.
Applications
Synthesizing agents: TRIETLENEGLYCOL can be used as synthesis intermediates. Triethyleneglycol esters containing fatty acids (oleic, stearic, lauric, etc.) are used as emulsifiers and plasticizers of polymers.
Brake fluids: TRIETİLENEGLIKOL can be used as a secondary solvent in brake fluid formulations. This product also prevents excessive rubber swelling in the hydraulic system.
Other uses: Ethyleneglycols can also be used in the formulation of printing ink, the treatment of gases, the formulation of fire-resistant hydraulic fluids, the formulation of cutting oils, the formulation of surface polishes, and the formulation of agrochemicals. , in the extraction of solvents, in the manufacture of pigmented pastes and pastes for walls, and the synthesis of explosives.